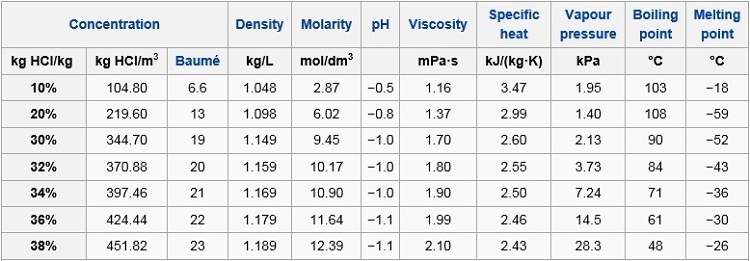
Effect of acid concentration on the oxidation of R·HCl. Acidity of the... | Download Scientific Diagram
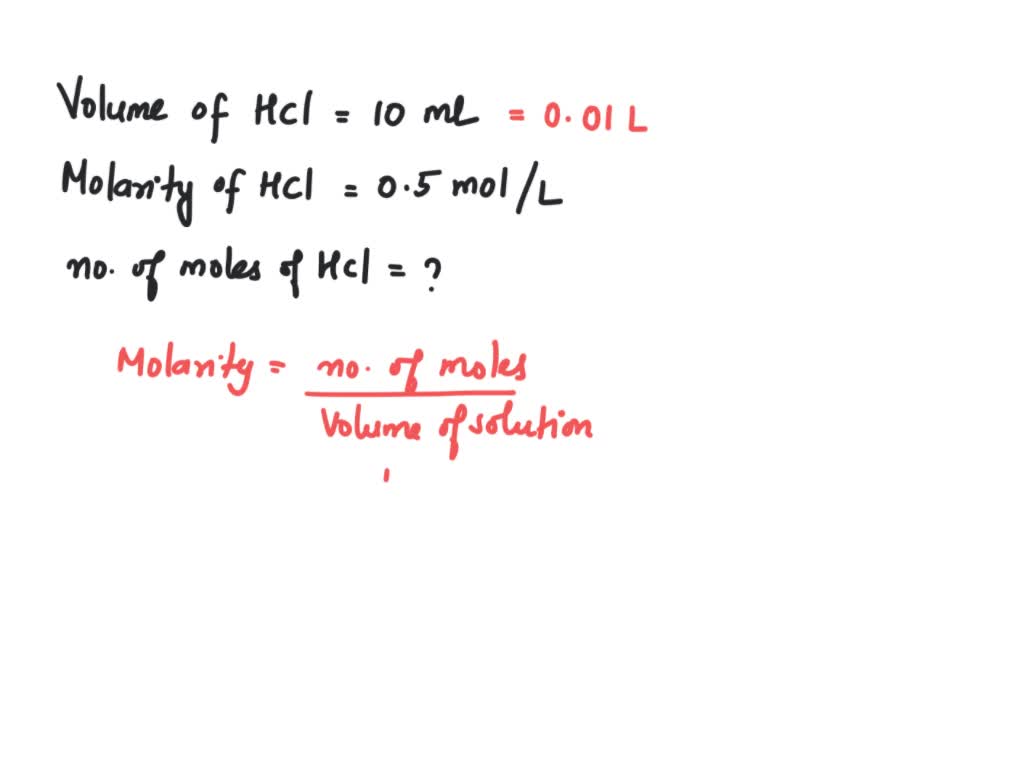
SOLVED: 1 How many moles of HCl are there in 10 mL of a solution with a concentration of 0.5 molL-I? swered Select one: 5 mole 0.05 mole of 200 Jestion 50 mole 0.5 mole

SOLVED: 1 How many moles of HCl are there in 10 mL of a solution with a concentration of 0.5 molL-I? swered Select one: 5 mole 0.05 mole of 200 Jestion 50 mole 0.5 mole
Hydrochloric acid, 500 ml, glass | Reagents for Decalcification | Reagents for Histology | Histology/Microscopy | Life Science | Carl Roth - International

A piece of metallic iron (10 moles) was dissolved in concentrated hydrochloric acid. The reaction formed hydrogen gas and iron chloride. How many grams of HCl were consumed? Don't forget the units. {