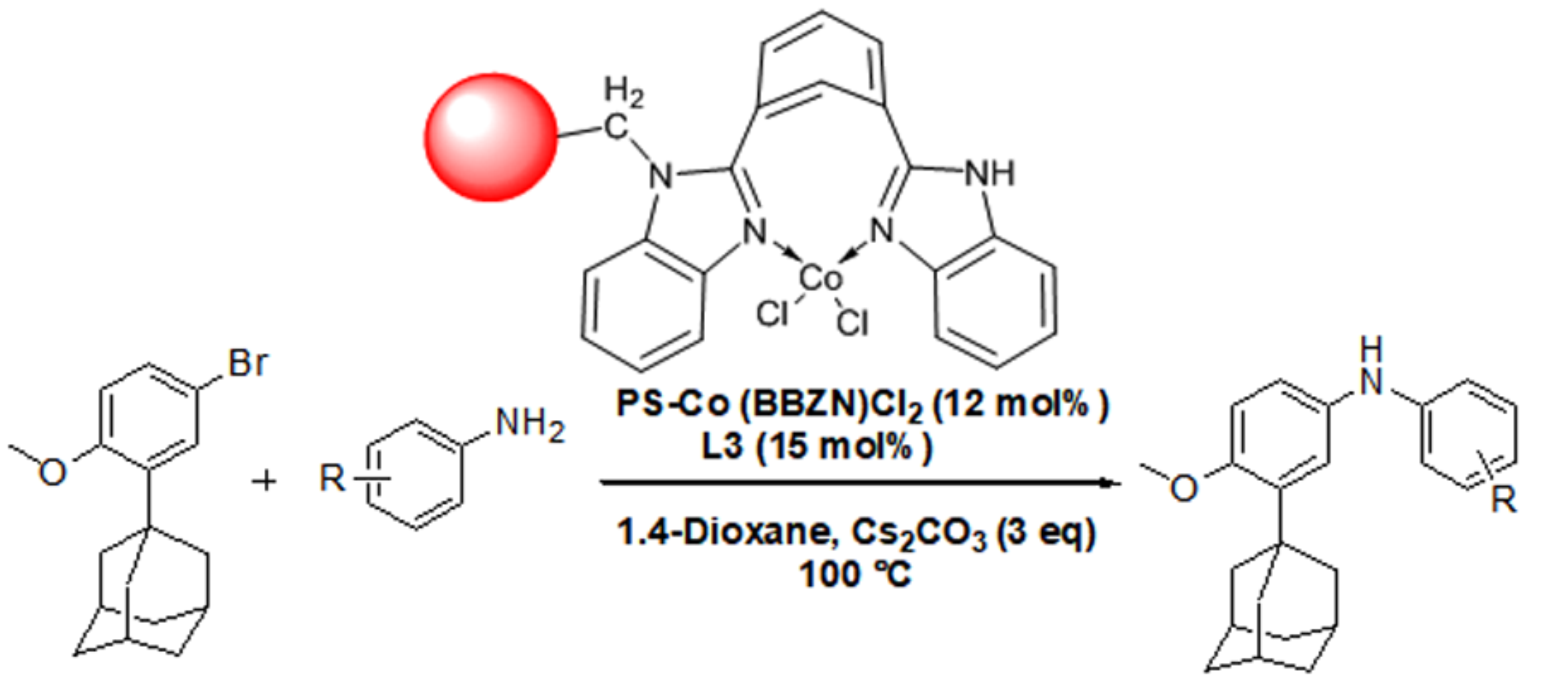
Catalysts | Free Full-Text | Development of a New Arylamination Reaction Catalyzed by Polymer Bound 1,3-(Bisbenzimidazolyl) Benzene Co(II) Complex and Generation of Bioactive Adamanate Amines

XRD pattern of (a) pure PVP and PVP doped with (b) 10 mol% (c) 15 mol%... | Download Scientific Diagram

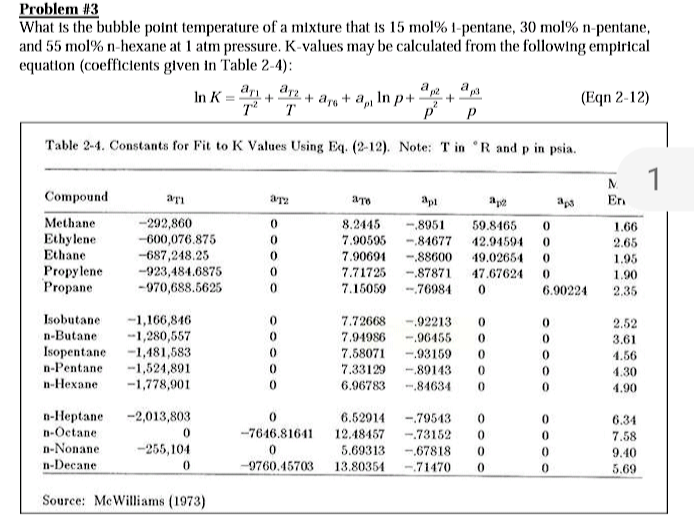
For, A + B C , the equilibrium concentration of A and B at a temperature are 15 mol litre^-1 . When volume is doubted the reaction has equilibrium concentration of A

Efficient and Recoverable Bio-Organic Catalyst Cysteine for Synthesis, Docking Study, and Antifungal Activity of New Bio-Active 3,4-Dihydropyrimidin-2(1H)-ones/thiones Under Microwave Irradiation | ACS Omega

Copper‐Catalyzed C(sp3)‐Amination of Ketone‐Derived Dihydroquinazolinones by Aromatization‐Driven C−C Bond Scission - Lv - 2023 - Angewandte Chemie International Edition - Wiley Online Library

りん酸緩衝剤粉末 (1/15 mol/L pH 7.4)・Phosphate Buffer Powder (1/15 mol/L, pH 7.4)・167-14491【詳細情報】|【ライフサイエンス】|試薬-富士フイルム和光純薬
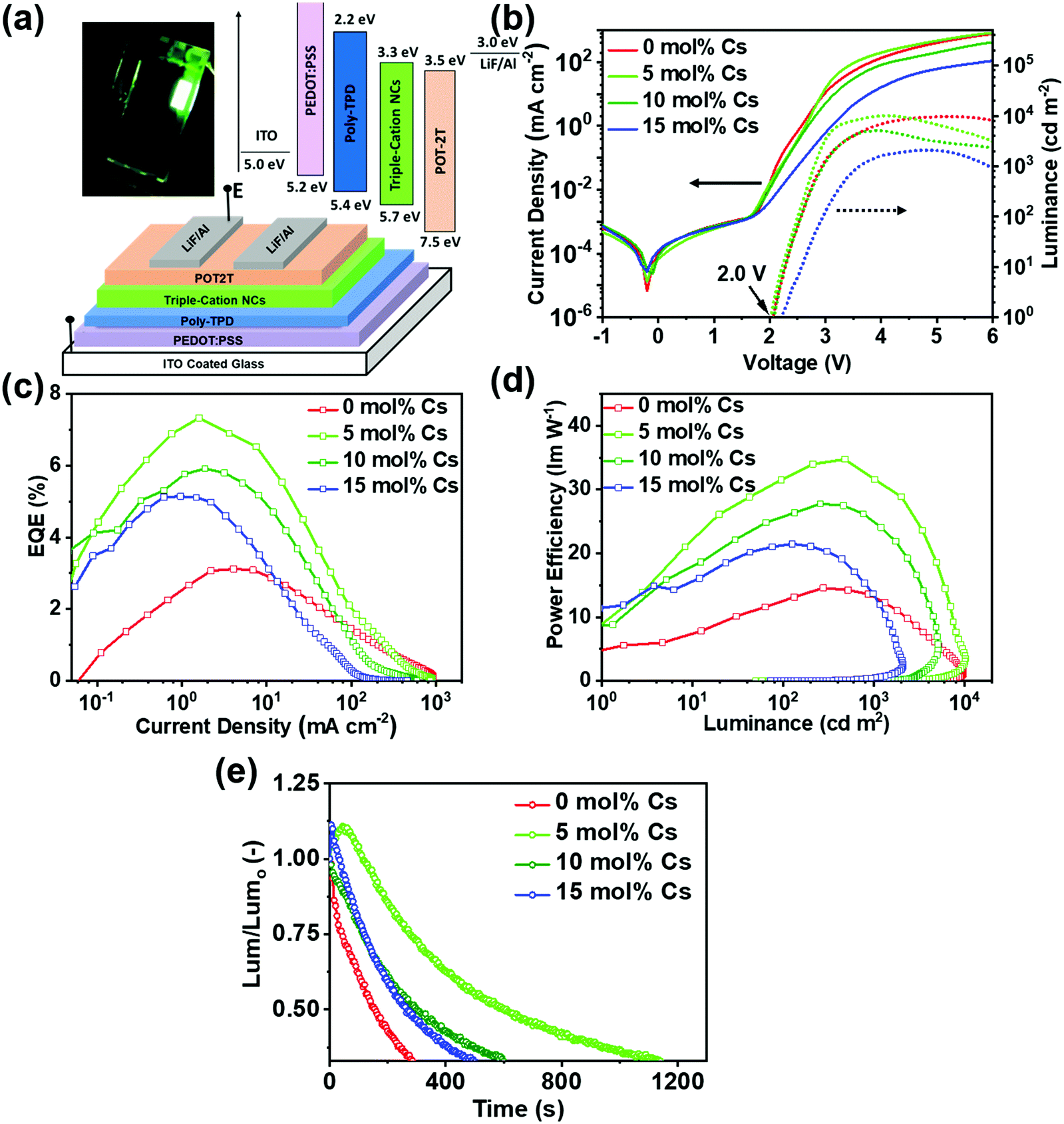
Investigating the structure–function relationship in triple cation perovskite nanocrystals for light-emitting diode applications - Journal of Materials Chemistry C (RSC Publishing) DOI:10.1039/D0TC02038A

SOLVED: if 0.40 mol of h2 and .15 mol of o2 were to reat as completely as possible to produce h20, what mass of the reactant would remain

Intracellular Communication between Synthetic Macromolecules | Journal of the American Chemical Society

Estimate the melting point range for a mixture of 85 mole percent phenol/15 mol percent diphenylamine - Brainly.com