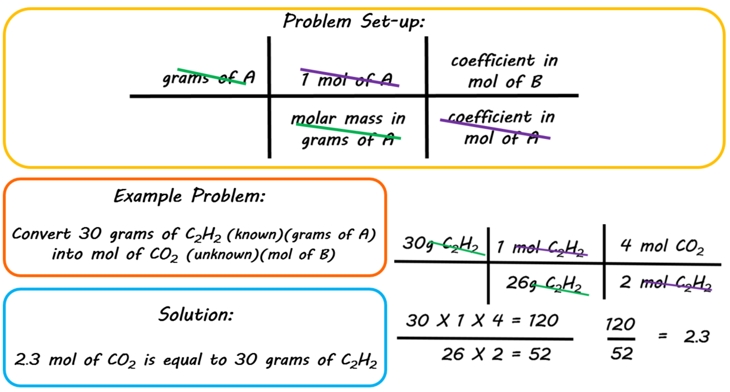
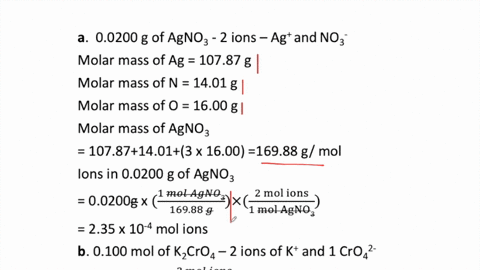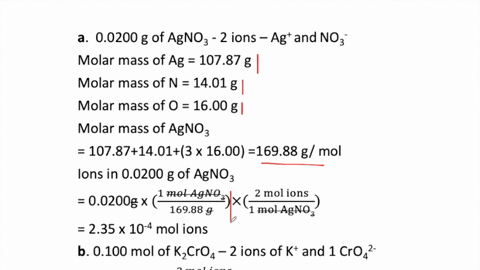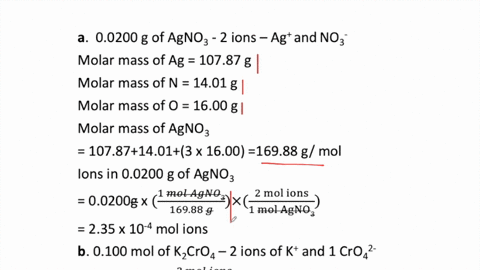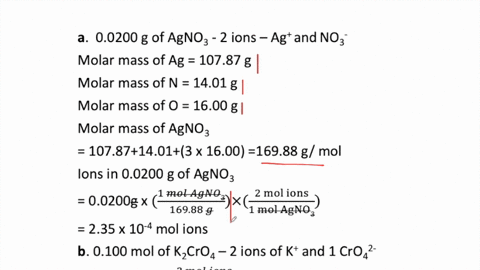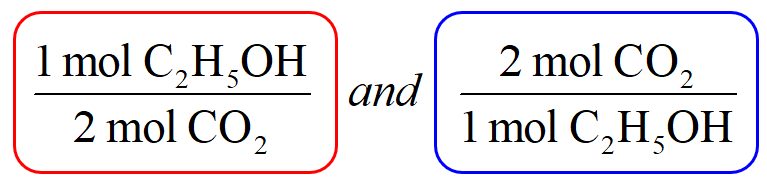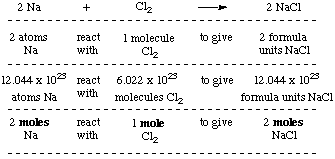2 moles of an ideal gas at temp 27 degree c is heated isoermall from volume v to 4v .if R=2 cal/mol then the heat input in the process is approximately

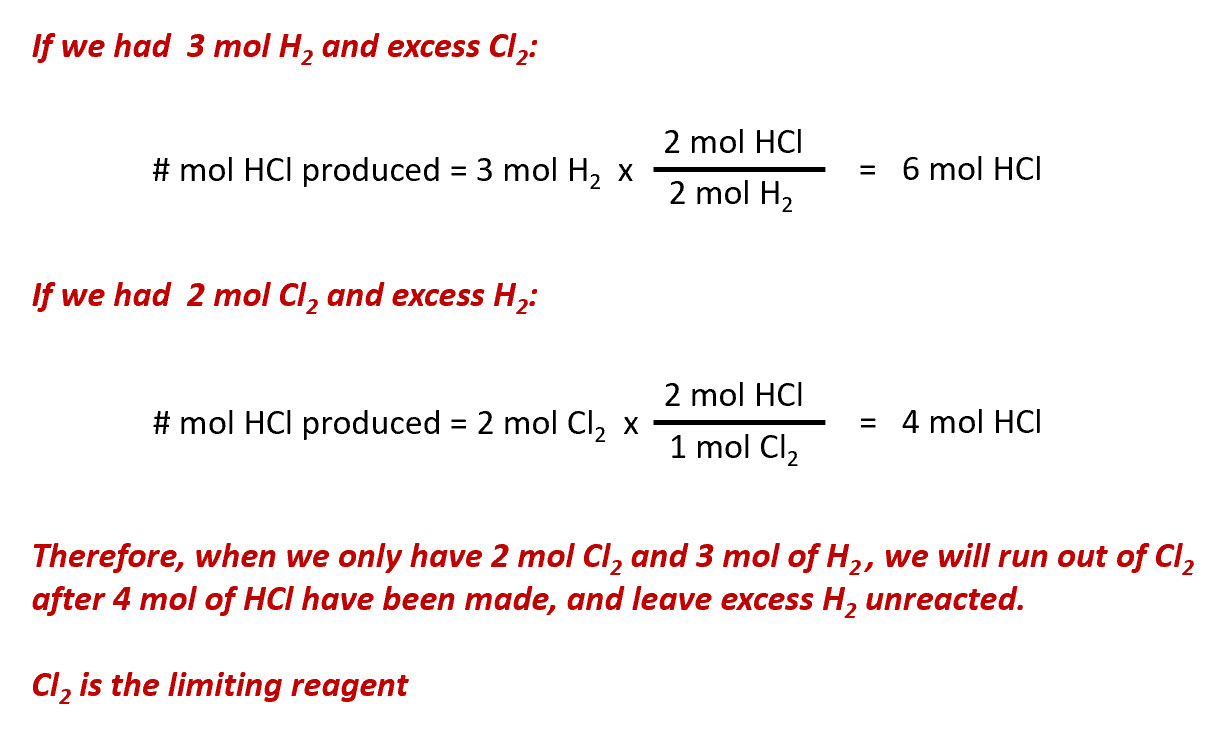
1 mole of N2 and 2 moles of H2 are allowed to react in a 1 dm^3 vessel. At equilibrium 0.8 mole of NH3 is formed. The concentration of H2 in the vessel is