When h+ forms a bottle of h2o to form the hydronium ion h3o plus this bond is called a coordinate - Brainly.com

Formation of Hydronium ion (H3O+) – Coordinate Covalent Bond Example - Best Online Free Chemistry Learning
![SOLVED: Which of the statements outlined below is NOI correct regarding the relationship between the hydronium and hydroxide ion concentrations, [H3O*] and [OH ], respectively, in relation to the pH of an SOLVED: Which of the statements outlined below is NOI correct regarding the relationship between the hydronium and hydroxide ion concentrations, [H3O*] and [OH ], respectively, in relation to the pH of an](https://cdn.numerade.com/ask_images/2c1d38b061434304a23e9db2b01d94f0.jpg)
SOLVED: Which of the statements outlined below is NOI correct regarding the relationship between the hydronium and hydroxide ion concentrations, [H3O*] and [OH ], respectively, in relation to the pH of an




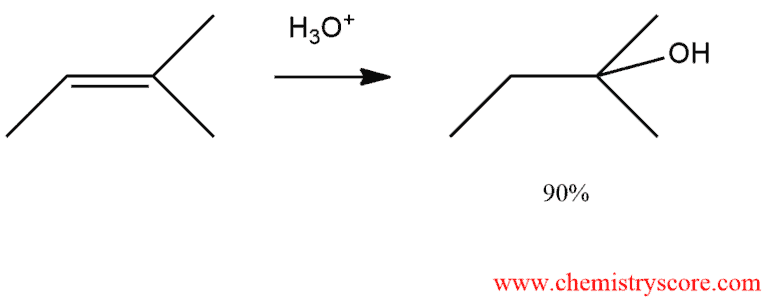
![Acetal hydrolysis [H3O+] - ChemistryScore Acetal hydrolysis [H3O+] - ChemistryScore](https://chemistryscore.com/wp-content/uploads/2019/11/Acetal-hydrolysis1-768x245.png)