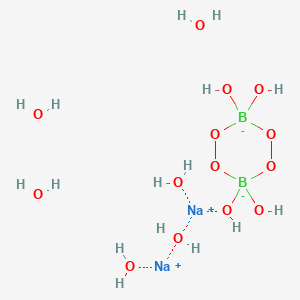
![PDF) Synthesis and Crystal Structure of [Na2(μ H2O)(H2O)CB[5]]Cl2 · 6H2O, [Na3(μ H2O)4(H2O)4(CNPy@CB[6])]Cl3 · 8H2O, and [Rb2(μ] H2O)2(CNPy@CB[6])]Cl · 8H2O | Ekaterina Kovalenko - Academia.edu PDF) Synthesis and Crystal Structure of [Na2(μ H2O)(H2O)CB[5]]Cl2 · 6H2O, [Na3(μ H2O)4(H2O)4(CNPy@CB[6])]Cl3 · 8H2O, and [Rb2(μ] H2O)2(CNPy@CB[6])]Cl · 8H2O | Ekaterina Kovalenko - Academia.edu](https://0.academia-photos.com/attachment_thumbnails/31549052/mini_magick20190426-32177-1j03o76.png?1556271006)
PDF) Synthesis and Crystal Structure of [Na2(μ H2O)(H2O)CB[5]]Cl2 · 6H2O, [Na3(μ H2O)4(H2O)4(CNPy@CB[6])]Cl3 · 8H2O, and [Rb2(μ] H2O)2(CNPy@CB[6])]Cl · 8H2O | Ekaterina Kovalenko - Academia.edu

Chemical Reactions Balancing Chemical Reactions. Chemical Reactions Objectives List three observations that suggest that a chemical reaction has taken. - ppt download
![Synthesis, crystal structure, thermal, photoluminescent and magnetic properties of a new material: Na2[Ni(C2O4)2(H2O)2].6H2O - ScienceDirect Synthesis, crystal structure, thermal, photoluminescent and magnetic properties of a new material: Na2[Ni(C2O4)2(H2O)2].6H2O - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0022286018311967-fx1.jpg)
Synthesis, crystal structure, thermal, photoluminescent and magnetic properties of a new material: Na2[Ni(C2O4)2(H2O)2].6H2O - ScienceDirect

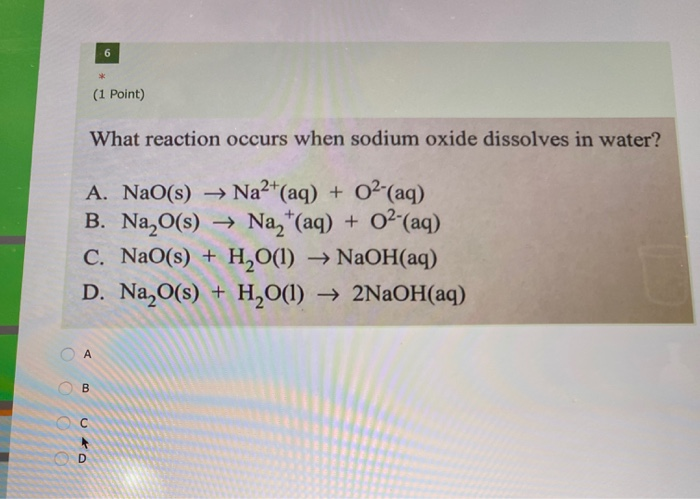
SOLVED: Use the following balanced chemical equation to answer this question: 2 Na + 2 H2O 72 NaOH + H2 How many pounds of hydrogen should be produced when 19.40 grams of
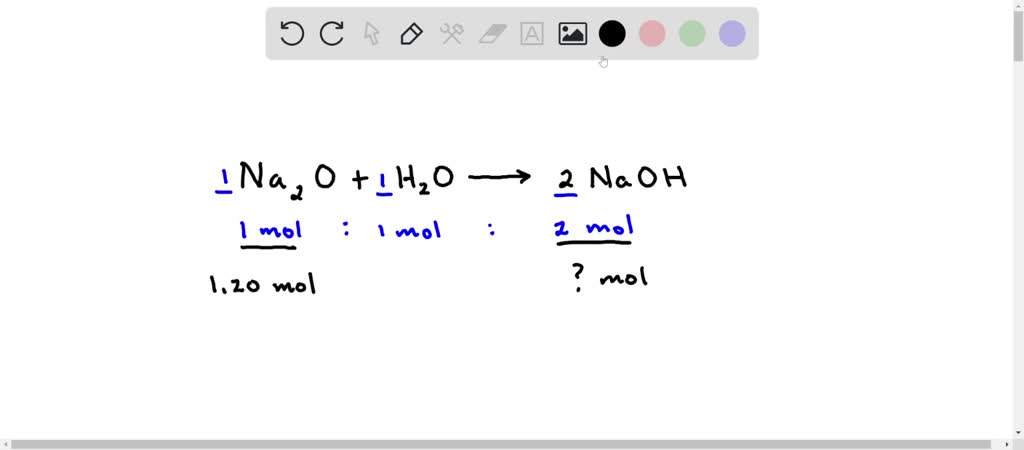
SOLVED: Given the following equation: Na2O + H2O —> 2 NaOH How many moles of NaOH are produced from 1.20 moles of Na2O?
![A novel blue luminescent material Na2[Co(C2O4)2(H2O)2]·6H2O: synthesis, structure, luminescence and magnetic properties - Dalton Transactions (RSC Publishing) A novel blue luminescent material Na2[Co(C2O4)2(H2O)2]·6H2O: synthesis, structure, luminescence and magnetic properties - Dalton Transactions (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/C6DT01185C)
A novel blue luminescent material Na2[Co(C2O4)2(H2O)2]·6H2O: synthesis, structure, luminescence and magnetic properties - Dalton Transactions (RSC Publishing)


![a) Exemplary structure of the complex Na2[Hf2(dpta)2] 7.5H2O .... | Download Scientific Diagram a) Exemplary structure of the complex Na2[Hf2(dpta)2] 7.5H2O .... | Download Scientific Diagram](https://www.researchgate.net/publication/352533926/figure/fig2/AS:1036268796379136@1624077142150/a-Exemplary-structure-of-the-complex-Na2Hf2dpta2-75H2O-05EtOH-b-2D-1-H-13-C.png)




![Synthesis and Crystal Structure of a New 3D Copper B-Paradodecatungstate Compound: [Na2(H2O)8][ Synthesis and Crystal Structure of a New 3D Copper B-Paradodecatungstate Compound: [Na2(H2O)8][](https://www.degruyter.com/document/doi/10.1515/znb-2009-0303/asset/graphic/s64b0269.gif)