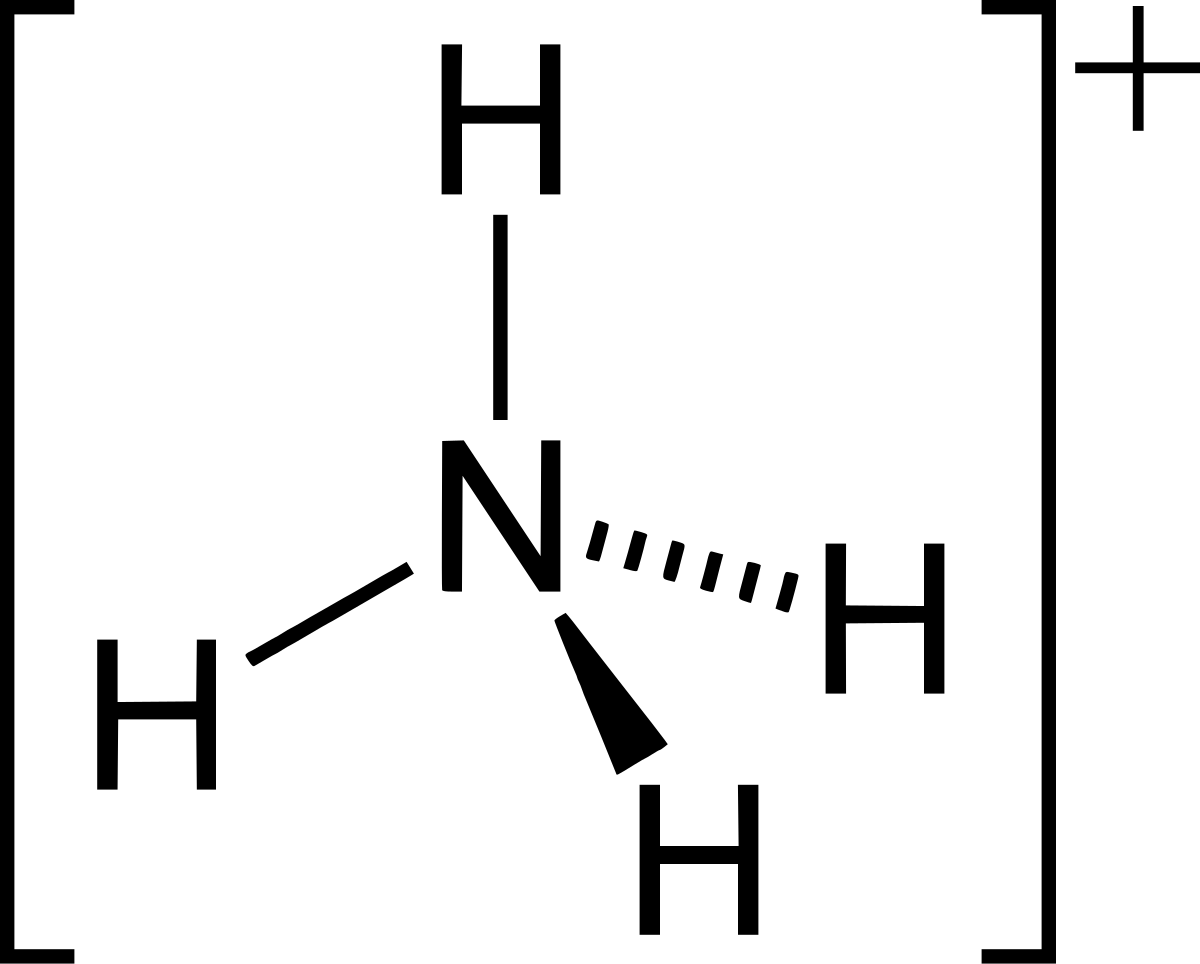
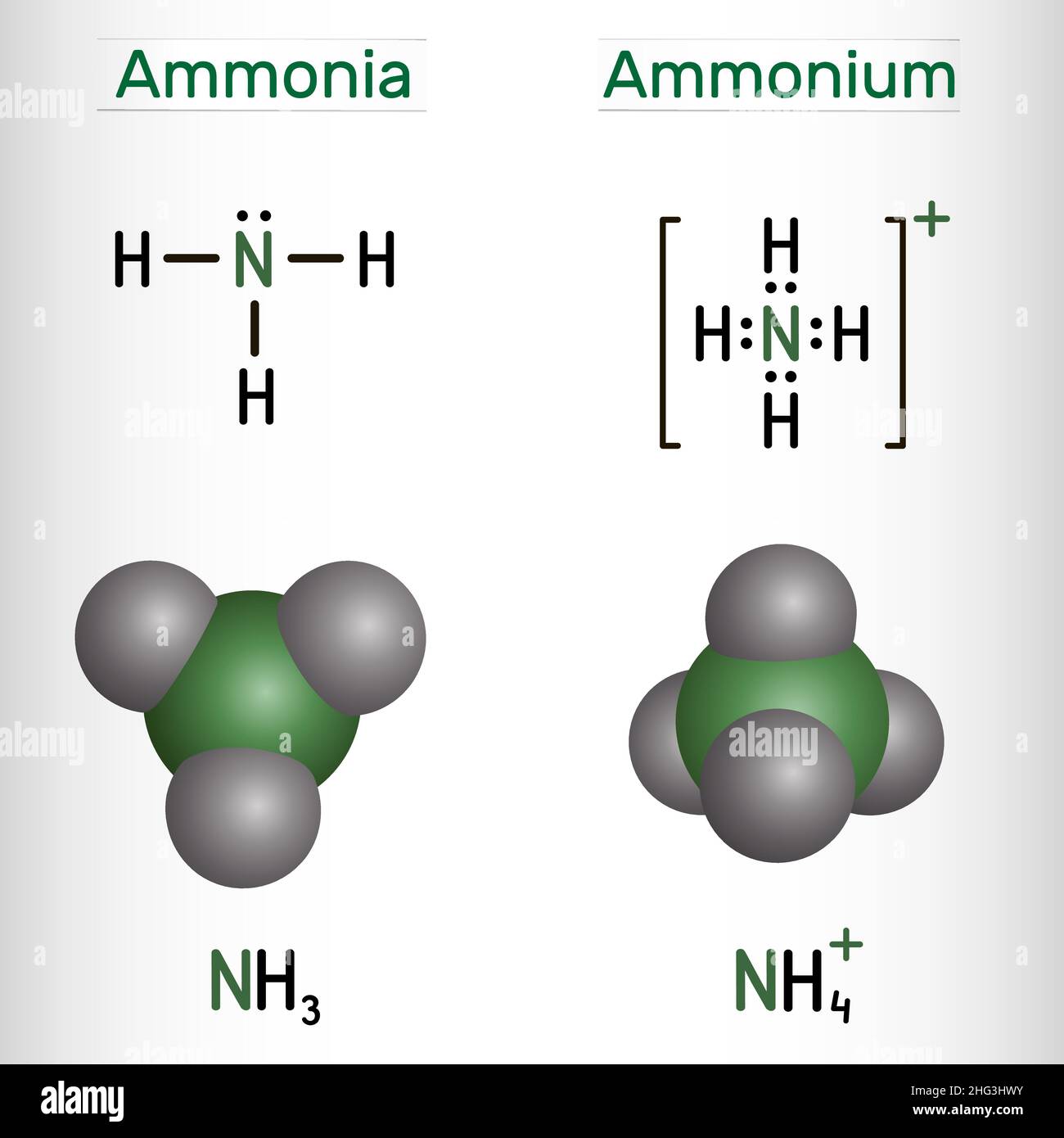
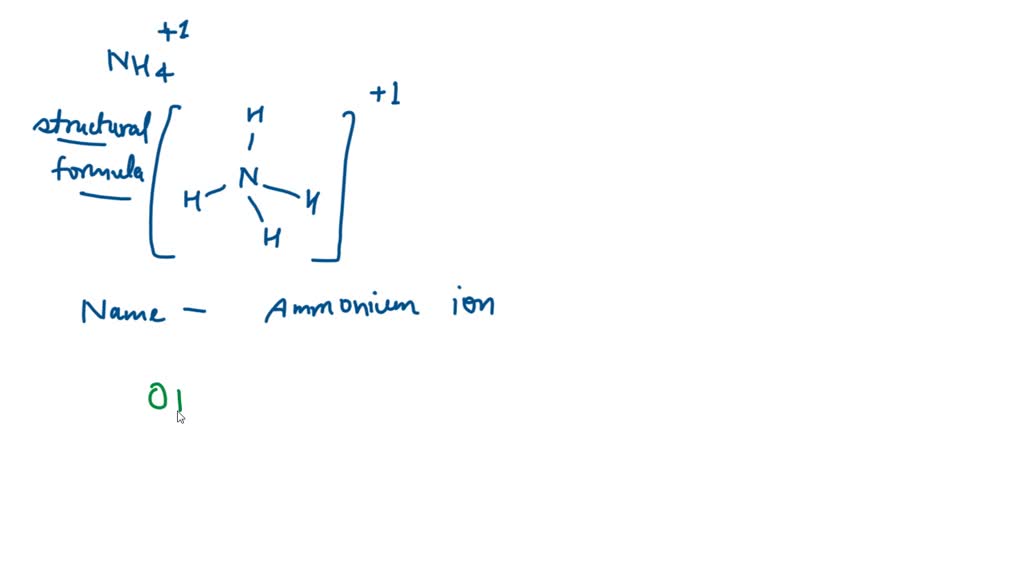
![grade 10: chemistry] Why is the number of moles of (NH4)2SO4 is half of the number of moles of NH3??? : r/HomeworkHelp grade 10: chemistry] Why is the number of moles of (NH4)2SO4 is half of the number of moles of NH3??? : r/HomeworkHelp](https://preview.redd.it/grade-10-chemistry-why-is-the-number-of-moles-of-nh4-2so4-v0-05mvin0yijl81.png?auto=webp&s=a504d96cf0488b87877861eba70cb1df0e9fcb75)
grade 10: chemistry] Why is the number of moles of (NH4)2SO4 is half of the number of moles of NH3??? : r/HomeworkHelp

Ammonium ion NH4+ - Explanation, Properties of Ammonium ion, Preparation and some Applications along with FAQs

6009-70-7 | Ammonium Oxalate Monohydrate | Ammonium Oxalate ((NH4)2C2O4) Monohydrate; | C₂H₂O₄ • 2H ₃N • H ₂O | TRC