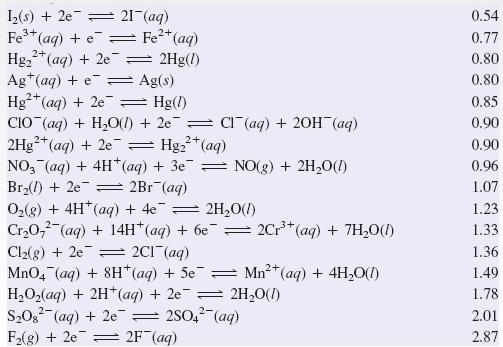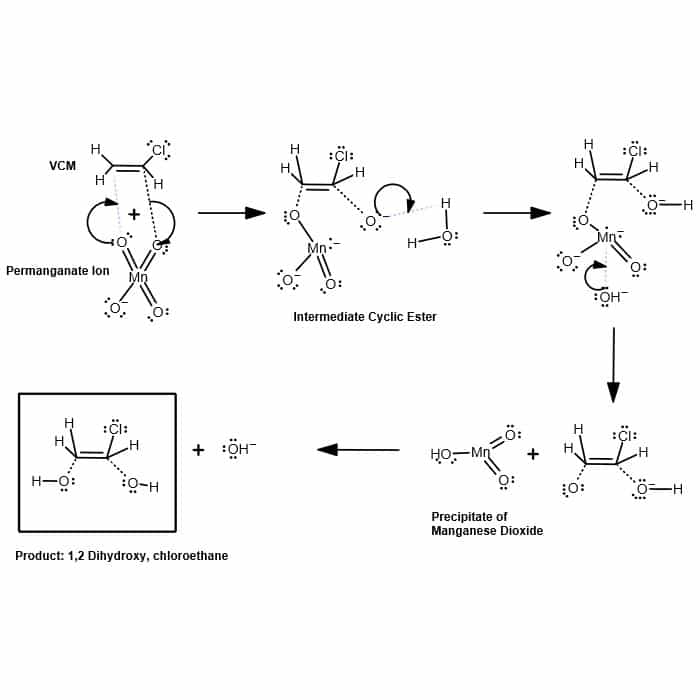
Reaction For Removal of Vinyl Chloride Using Potassium Permanganate | HS-600 | Hydrosil International

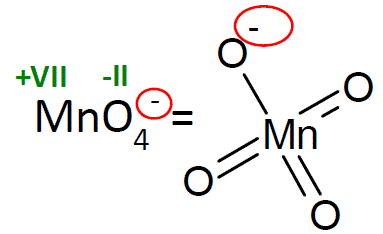
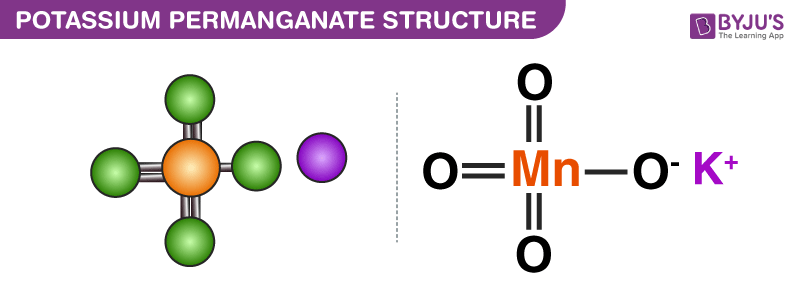
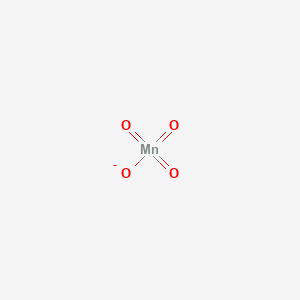
SOLVED: Section 2 The concentration of oxalate ion in a sample can be determined by titration with a solution of permanganate ion of known concentration. The net ionic equation for this reaction
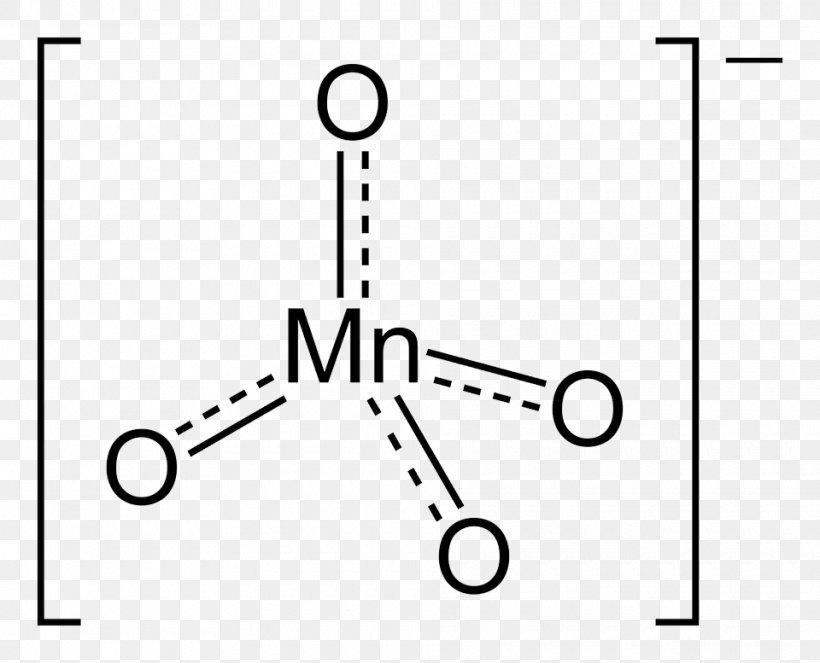
Potassium Permanganate Lewis Structure Ion, PNG, 960x777px, Watercolor, Cartoon, Flower, Frame, Heart Download Free
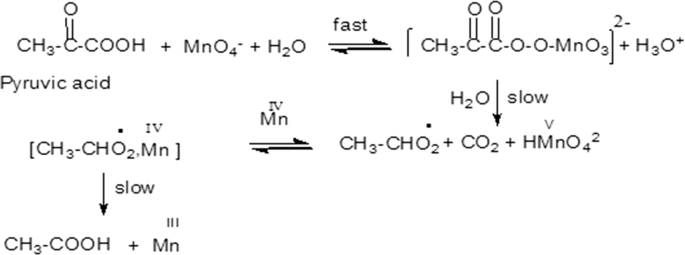
Kinetics and mechanism of oxidation of pyruvate by permanganate ion in aqueous perchlorate solution | SpringerLink

Manganat-Ionen disproportionieren zu Braunstein und Permanganat-Ionen - Reaktionsgleichung | Philus Social eLearning -
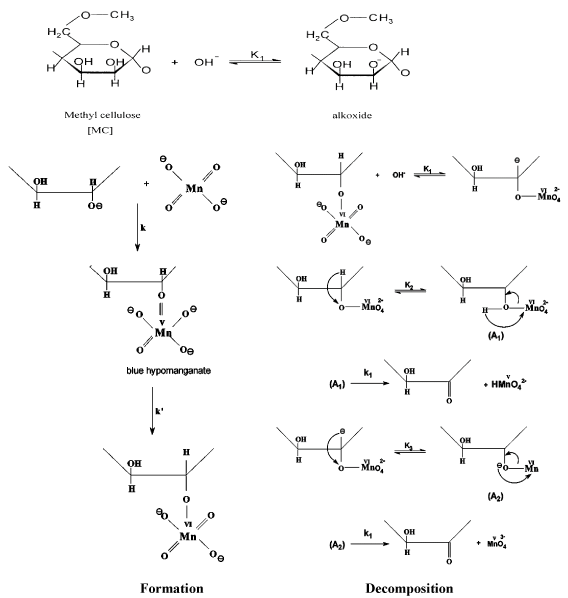
Novel synthesis of coordination biopolymer precursor from oxidation of methyl cellulose by alkaline potassium permanganate
How is a permanganate ion negatively charged and still capable of accepting electron in reduction as it is an oxidizing agent? - Quora